Market Release Rates Reveal Shortages of Imported Veterinary Medicines
Imports of veterinary medicinal products have been slowly recovering following the changes in market access conditions. The process has been slowed down by long GMP audits procedure, the difficult resumption of production after being GMP certified and the delivery of necessary reagents.
AVPHARM has analyzed data on imported veterinary medicinal products released to the market from September 2023 to June 2024. Over the entire period since the new procedure was put in place last September, foreign manufacturers released to the market 164 VMPs. The list encompasses 97 veterinary pharmaceuticals and 68 vaccines, out of which the majority (45) are for use in poultry, 12 are swine vaccinations, and 11 are for cattle and/or small ruminants. Notably, all the imported vaccines are produced by six companies. The data is presented in the line graph below.
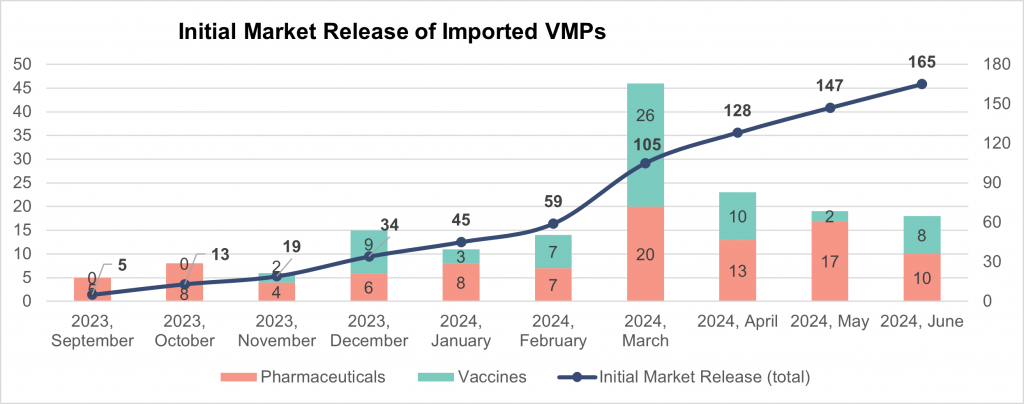
Last summer, AVPHARM predicted that with the introduction of the market release procedure in September 2023, the Russian market would import between 160 and 180 types of VMPs. Throughout the entire period, the rate of initial market release remained low, and it was not until June 2024 that the predicted range of products finally became available to customers. It is worth mentioning that in 2022, 750 veterinary medicinal products, including 220 vaccines, were delivered to the country.
Semyon Zhavoronkov, Executive Director of AVPHARM, dubbed the current situation both “predictable and, unfortunately, predicted.”
“Back in 2019, the industry-related business, the public and even the Ministry of Economic Development highlighted the risks of a significant shortfall in supply of foreign pharmaceuticals and vaccines for animals. Although the rules, effective since September 1, were just being conceived at that time, the problem was the same: global animal health companies lacked Russian GMP certificates that were necessary at first for the registration of their products and now for the delivery of their already registered medicines. As of now, more companies have been GMP certified, and some progress has been made in terms of positive inspection outcomes. However, the momentum must be maintained to ensure greater availability of foreign veterinary medicinal products in the local market,” he said.
The March and April surge in the number of veterinary medicinal products, primarily vaccines, which were released to the market is correlated with the major manufacturers obtaining Russian EAEU GMP certificates in December and January. In fact, the data confirms that if a company was forced to suspend the production of its veterinary medications intended for the Russian market by September 2023, it will take a lot of time to resume it. The GMP audit produces unpredictable outcomes, which is why only after being certified can one place market orders. The time when VMPs become available for sale depends on many factors, but it takes four or eight months if the conditions are favorable.
Along with placing production orders, the companies arrange for particular materials – quite often for each veterinary medicinal product – to be gathered and delivered, with those including testing kits and laboratory reagents and consumables. The listed products must be delivered to comply with quality control standards that are part of the rules governing Russia’s market release procedure. Despite the fact that the testing period is subject to the relevant regulations, it may take more than six months to approve and deliver all the necessary materials under the effective legal framework. Moreover, given the export control requirements, some reagents cannot be shipped to Russia at all.